Features
Crop Protection
Inputs
Narrowing down your pesky pest problems
Insect and disease issues can be hard to diagnose correctly. These tips can help you save time and money by arriving at the culprit sooner.
September 10, 2019 By Dr. Sarah Jandricic
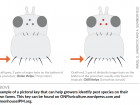
 Uniform damage throughout the crop and on leaves of all ages within the plant suggests an abiotic problem. Here, phytotoxicity is pictured and not a disease. Photo: OMAFRA
Uniform damage throughout the crop and on leaves of all ages within the plant suggests an abiotic problem. Here, phytotoxicity is pictured and not a disease. Photo: OMAFRAWe all know what they say about assumptions. When diagnosing crops with little tolerance for damage, it’s important not to let preconceived notions lead you down the wrong path. Here are some common mistakes to avoid making when diagnosing insect and disease issues in ornamentals.
1. Not Asking the Right Questions
There are some important questions to ask yourself before you start to narrow down the possible offenders. These questions let you look at the issue objectively and pick the best fit from production guides or extension web pages, rather than subconsciously making your problem “fit” the description. Additionally, having this information handy speeds up the process if you turn to a consultant or specialist for further help.
- Which crop? Does it affect multiple crops in one area? Or just a specific cultivar? More than one crop or variety suggests a widespread problem like flying insects, or an airborne disease with a wide host range (e.g. powdery mildew, Botrytis). A single variety affected amongst an otherwise healthy crop often points to greater susceptibility to a virus or phytotoxicity.
- When did it start? Some diseases are likely to follow environmental events (e.g. Botrytis outbreaks after a wet, cloudy period). Similarly, damage from certain insects can show up around the same time each year (e.g. damage on poinsettia from Lewis mites in October). But damage that appears “overnight” can point to something other than insects or disease.
- How is the damage distributed? Clumps of damage within a crop could suggest slow-moving insects (e.g. mites, aphids, onion thrips) or diseases spread through contact (bacterial leaf spots). Random, patchy distributions can suggest a slow-developing disease that started on injured cuttings (e.g. Fusarium wilt). Patterns within the plants themselves will also give you more clues. Some pests prefer new growth (e.g. broad mite), while certain diseases girdle branches, causing wilt on one side of the plant (e.g. Botrytis stem blight).
- What have you changed? Growers are often hesitant to admit that changes in their production practices could cause damage, but we see this more often than you think. Shade curtains not closed fast enough on a sunny day, changes in media that affect water needs, and use of a pesticide on a new variety can all result in leaf burn that looks like insect or disease damage. Before you jump to conclusions about pests, make a list of changes made since the crop was last grown, and note how these could have impacted the crop. An issue that presents with an even distribution throughout the crop or affects a high percentage of plants (i.e. >60%), almost always ends up being an abiotic issue (i.e. nutritional, environmental or phytotoxicity) rather than biotic (e.g. insect, disease).
2. Not Taking Diagnostics Far Enough
Though many foliar diseases (e.g. powdery mildew, downy mildew, common viruses) can be diagnosed with a hand lens, a guide book, and some experience, anyone who tells you they can positively ID a root rot based on symptoms alone might also try to sell you a bridge before they leave your greenhouse.
For example, Phytophthora crown rot cannot be positively distinguished from Pythium aphanidermatum injury in poinsettia. Both cause similar-looking root issues that can extend up the lower stem. Suspected root diseases should always be confirmed with lab DNA analyses. Since not all fungi that show up on DNA reports may be pathogenic to ornamentals, do some research or have a consultant interpret the results to know what’s worth targeting with management tactics.
Similarly, it’s important to know exactly what insect you’re facing, especially if you’re suddenly having trouble controlling it. Recently, some Ontario growers thought their biocontrol programs for Western flower thrips were failing. Once the outbreak was properly identified as onion thrips (Thrips tabaci), the appropriate management tactics could be used. OMAFRA and Vineland Research and Innovation Centre are developing easy-to-use grower identification keys for key pests such as thrips, aphids and mites to make on-farm identification easier.
3. Ignoring Test Results
I’ve seen it more than once: a grower struggling with widespread damage and a negative DNA report from a lab, insisting it has to be disease. This reaction is understandable; diseases can be fixable— unlike many other production issues. But it’s important to keep emotion out of it and follow the evidence.
The chances of molecular tools, like a DNA multiscan analysis, sending back a false negative from a good sample of symptomatic tissue is low. This is true even after treatment (e.g. chemical fungicides) since molecular techniques detect the presence of both live and dead DNA.
One way to help ensure that your lab test is accurate (or at least more convincing) is to send multiple plants so the lab can pick the ‘“best” sample. You can also see if the lab can grow a sample of the suspected disease on bacterial- or fungal-specific media. This can act as a confirmation for the DNA results: if nothing grows from the sample when given its favourite food, there was never likely a disease there in the first place.
If you’ve followed these steps and your pathogen tests are still negative, then it’s time to investigate production issues that might have led to injury. Environmental problems are more common than you think. For example, cut flower greenhouses kept too hot can see weakened, hollow stems that mimic bacterial infections. Certain pesticides can have delayed or unexpected phytotoxicity, especially when applied as a drench or on open flowers. Even PGRs can damage a crop, if applied using a boom that’s left dripping over a part of the crop, for example. Look back through your spray and temperature records to help match up the time period when damage appeared.
4. Not Having the Right Tools
Let’s be honest: it’s hard to see many key insect characteristics (e.g. antennal length in aphids) with only a 10-15X hand lens. Signs of disease (hyphae, spores) are even harder. One of the best tools for fast, accurate, in-house diagnostics is a microscope.
Buying a microscope might sound intimidating, but for less than $500, you can find one that will get the job done for years to come. Look for these features:
- Maximum magnification of at least 45X. To determine this, take the highest number on the adjustable magnification dial (e.g. 4.5) and multiply it by the magnification of the eyepiece (usually 10X) given in the description.
- A light source. Most microscopes have this built in, but you’ll want to make sure it’s got both a top and bottom light source to aid with different views.
- Make sure it’s binocular (has two eye pieces) and is forward mounted (the eyepieces are on the same side as the stage). Other designs are very frustrating to use.
- A good USB camera of at least 16 megapixels is a bonus – this way you can share images internally or send them to a consultant or specialist for faster diagnosis.
With these tips in mind, along with your notes made each time you encounter a specific issue (and what resolved it), diagnosing and eventually preventing pests and diseases will become easier with time.
Sarah Jandricic, PhD, is the greenhouse floriculture IPM specialist for the Ontario Ministry of Agriculture, Food and Rural Affairs. She can be reached at
sarah.jandricic@ontario.ca.
Print this page